|
Aromatic Compounds with Multiple Substituents For instance, an aromatic ring bonded to an 8-carbon chain would be 1-phenyloctane, and not octylbenzene. If the substituent contains more than six carbons, the alkane portion is named first, and the aromatic ring portion is added as a suffix. Alkyl groups are named according to the alkane series convention ending with -yl: methyl (for a single carbon), ethyl (for two carbons), propyl (for three carbons), etc. When there is a single substituent on a benzene ring and the substituent contains six or fewer carbons, the substituent is included as a prefix to benzene. Aromatic Compounds with a Single Substituent Naming aromatic compounds : This nomenclature tutorial video takes you through the IUPAC rules for benzene type molecules and includes the common names for substituted benzene. For example, methylbenzene is often referred to as toluene, and dimethylbenzene is often called xylene. These names are frequently used in favor of standardized IUPAC nomenclature. There are a number of historically common names for aromatic structures. para: A prefix used to name an aromatic ring with two substituents directly across from one another on the ring.Īromatic compounds are ring structures with unusual stability due to delocalized pi electron density that is shared between all of the carbon atoms in the ring. 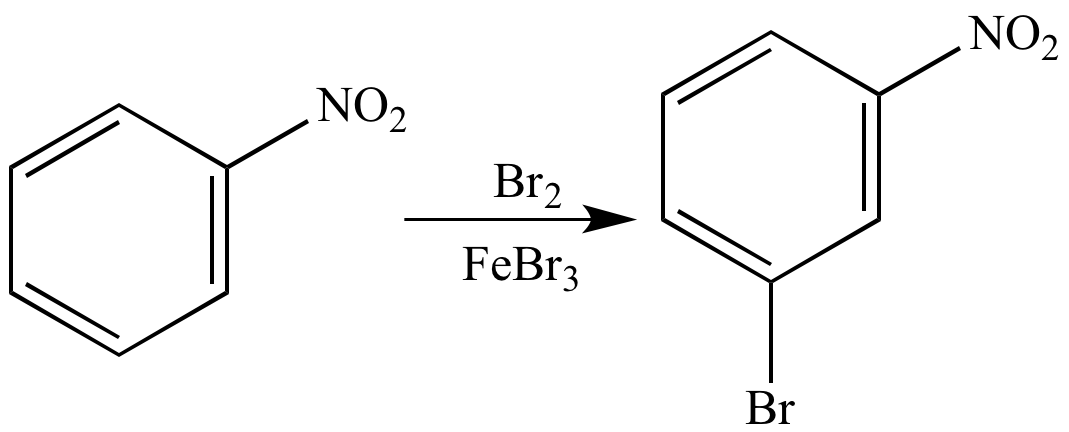  
meta: A prefix used to name an aromatic ring with two substituents separated by one carbon on the ring.ortho: A prefix used to name an aromatic ring with two adjacent substituents.For benzene rings with multiple substituents, the ring atoms are numbered to minimize the numbering of the substituents groups alternatively, ortho/meta/para nomenclature can be used for disubstituted rings.For substituted benzene rings where the substituent contains less than six carbons, the alkyl chain is added as a prefix with the ending changed to -yl.For substituted benzene rings where the substituent contains more than six carbons, the benzene ring is noted by using a phenyl prefix on the alkane name.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
